Medicine, Dentistry, and Pharmacy
April 28, 2023
Targeting mitochondria and related protein suggest new therapeutic strategy for treating Lou Gehrig's disease (ALS)
Researchers at Nagoya University in Japan have discovered a receptor, sigma-1 receptor, and a protein, ATAD3A, that are associated with Amyotrophic Lateral Sclerosis (ALS), also known as Lou Gehrig’s disease. Since there are drugs that specifically target the receptor, their findings suggest a new therapeutic strategy. They published the study in the journal Neurobiology of Disease.
ALS causes degeneration of motor neurons and the resulting muscle atrophy. Some of this degeneration is the result of the dysfunction of mitochondria, the energy-generating organelles of the body. This dysfunction causes a lack of energy in neurons resulting in the characteristic symptoms of the disease.
The integrity of the mitochondria-associated membrane (MAM) is important for the stability of the mitochondria. The MAM is especially important during the processes of division of mitochondria (called fission) and mitochondria fusing together (called fusion). Several proteins, including enzymes, are associated with these processes and accumulate in the MAM.
In patients with ALS, the mitochondria are damaged and undergo a process called fragmentation, which is associated with decreased cell fusion and increased fission. As cells increasingly divide, mitochondria break down into smaller organelles that have impaired function.
“Researchers have found this process of fragmentation in various neurodegenerative diseases,” said Professor Koji Yamanaka (he/him). “Discovering the detailed mechanism of how this process occurs would be useful to treat not only ALS but also other diseases.”
Since proteins rarely show activity on their own, to exert an effect, they often form assemblies, called dimers. A protein found in the MAM, called ATAD3A, is often found in dimers that cause mitochondrial dysfunction. Because these dimers have been found in diseases that cause nerve cells to die, such as Huntington’s and Alzheimer's, Yamanaka and his colleagues tested whether ATAD3A could also be involved in patients with ALS.
The group consisted of Professor Koji Yamanaka, Assistant Professor Seiji Watanabe (he/him), and graduate student Mai Horiuchi (she/her) at the Research Institute of Environmental Medicine at Nagoya University. They found that the interaction between ATAD3A and a receptor, called the sigma-1 receptor, contributes to the homeostasis of the MAM. When they tested models deficient in sigma-1 receptor–ATAD3A, they found they were associated with mitochondrial fragmentation.
“Loss of sigma-1 receptor function causes the inherited form of ALS,” said Yamanaka. “In healthy subjects, the sigma-1 receptor interacts with ATAD3A to suppress mitochondrial fragmentation. Both the sigma-1 receptor and ATAD3A are critical factors of MAM to maintain mitochondrial homeostasis, and we found that they are altered in the mouse model of ALS.”
Their findings also suggest potential treatments for patients with ALS. “Administration of a sigma 1 receptor agonist extended the survival time of ALS model mice,” Yamanaka explains. “Targeting sigma-1 receptor–ATAD3A at the MAM to prevent mitochondrial dysfunction would lead to the development of a novel therapeutic strategy for neurodegenerative diseases and may be a suitable target for future drug development for ALS.”
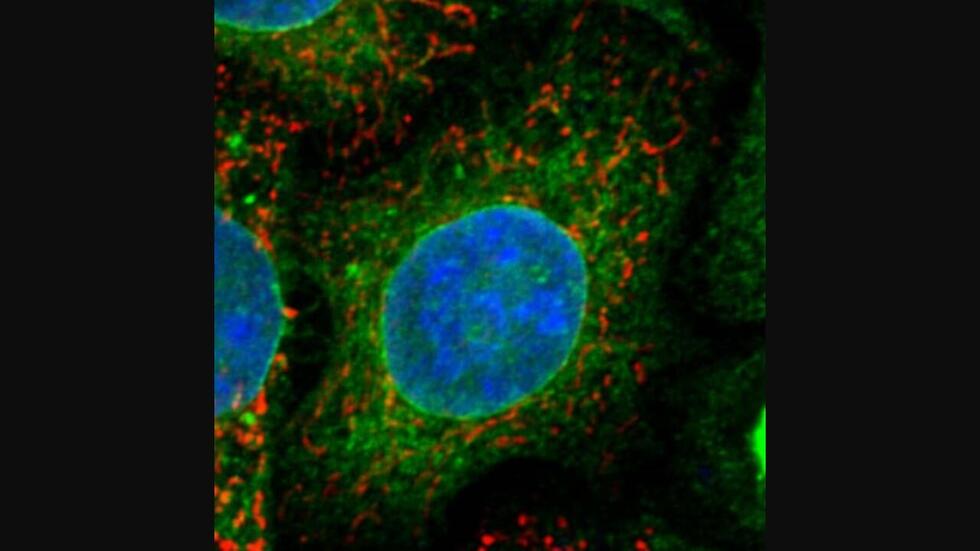
Researchers have discovered a receptor, sigma-1 receptor (green), and a protein, ATAD3A (red),
that are associated with Amyotrophic Lateral Sclerosis (ALS), also known as Lou Gehrig’s disease.
Image credit: Yamanaka Laboratory
The study, "Sigma-1 receptor maintains ATAD3A as a monomer to inhibit mitochondrial fragmentation at the mitochondria-associated membrane in amyotrophic lateral sclerosis," has been published in the journal Neurobiology of Disease at DOI: 10.1016/j.nbd.2023.106031.
Authors:
Seiji Watanabe, Mai Horiuchi, Yuri Murata, Okiru Komine1, Noe Kawade1, Akira Sobue, and Koji Yamanaka
Media Contact:
Matthew Coslett
International Communications Office, Nagoya University
kouho-en@adm.nagoya-u.ac.jp